Plasmonic PCR™
A revolutionary new PCR development leading to the potential for COVID-19 detection in under 1 minute
Interested? Please contact us for more information.

| Plasmonic PCR | Existing PCR | |
|---|---|---|
| Time | < 6 minutes (current) |
> 30 minutes |
| Labels | Label free real time monitoring of amplicon, sensitive to 10 dna copies |
Requires Labels |
| Sample Preparation | Automated, hands free, chemical free | Requires extensive sample preparation |
| Point of Care | Battery, portable | High power consumption, not portable |
| Safety | Inherent | Less Safe |
| Compatibility | Compatible with PCR, RT-PCR and LAMP formats | Standard Compatible |
| Channels | Up to 8 channels. Four channel system is shown |
Significance
The global need for point-of-care (POC) viral testing. The COVID-19 China outbreak and the previous outbreaks of related RNA viruses MERS and SARS have demonstrated that delays in diagnosis leads to non-optimal management of individuals infected with these highly pathogenic viruses. The present method for COVID-19 diagnostic testing is RT-PCR using conventional costly and workflow laborious qPCR apparatuses, performed in the central laboratory and being far from POC. This is because traditional thermocyclers typically employ electric heating blocks which are large and require excessive amounts of energy to heat and cool a Peltier block, making them 90% energy inefficient. This is compounded by the fact that the PCR tube represents only a fraction of the entire thermal mass that must be heated or cooled. The speed of heating and or cooling is rather limited. To meet this challenge, we have re-invented and patented a PCR methodology using a completely novel and exceedingly simple way to generate and manage heat needed for thermocycling.
In partnership with Drs. Mark Trifiro, Miltiadis Paliouras, and Andrew Kirk, we introduce the new paradigm in PCR diagnostics.
Device Description
1. Temperature cycling in seconds
Our technology is achieved using Gold Nanoparticles in the PCR reaction mixture itself. Specifically, Gold nanorods (GNRs) from Nanopartz when exposed to laser light can generate enormous heat in femtoseconds. By manipulating a low powered 808nm laser to turn on and off (cycling/second), heating is instantaneous and extremely precise. GNRs have exemplary light absorption properties (>90%) and are 100% efficient in converting absorbed light to heat reaching rates of 20°C/second. Temperature monitoring is by non-contact IR thermometry (up to 50 measurements/sec). Since the tube is completely free, not entombed into a block, cooling is accomplished by convection. Simple, efficient and extremely rapid thermocycling can be achieved, allowing for a highly specific PCR product (FIG 1A). It is also completely compatible with all existing PCR protocols, has very convenient 10-25 µl reaction volumes, and has the potential to operate at any scale (with the use of microbeam lasers a small scale is feasible). As the energy needs for our Plasmonic PCR are vastly reduced, a POC self-powered portable version can be manufactured.
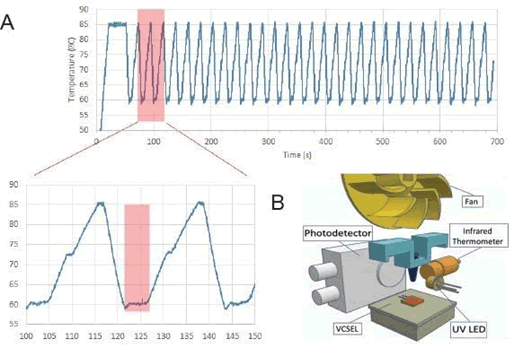
FIG 1. Plasmonic PCR. A. Shows an example of temperature monitoring of a 30-cycle Plasmonic PCR experiment. The plasmonic system maintains the stability of temperature over longer extension periods, see enlarge highlighted box. B. Schematic representation of VCSEL system, with UV-LED and photo- detector placement.
We have also optimized the parameters of the PCR. PCR stages are held for only 1-2 seconds (extension and denaturation). For the annealing step, currently 4 seconds works best and yields highly specificity amplicons than conventional PCR (FIG-1A). These times are ample to yield a reliable maximal >200bp PCR product. With our present Plasmonic PCR we complete a 30 cycle PCR in under ten minutes. Plasmonic PCR achieves excellent PCR efficiency with excellent yield and sensitivity.
2. Label Free Real Time Amplicon Quantization
Included in our Plasmonic PCR technology is real-time monitoring of amplicon production. In keeping with an all-optical approach, we have employed an innovative approach whereby no fluorescent label is required (i.e., label-free). This is achieved by assessing UV absorption properties during PCR cycles. Most recently, we have adapted the Plasmonic PCR to be able to monitor real-time fluorescence PCR, with Ct values indistinguishable from conventional qPCR platforms. These innovations significantly increase the scope of our Plasmonic PCR, completing and interpreting PCR in real-time.
![]()
FIG 2. UV Monitoring of DNA amplification. A. Plotted results of UV monitoring of different copy numbers of C. trachomatis DNA, from 10,000 to 10. UV monitoring, can confidently measure with certainty as low as 1,000 copies. B. Gel electrophoresis of corresponding experiment, shows the strength of the DNA amplicon at 10,000 copies. This would represent less than 1 CFU, although our UV monitoring detection is 10-fold more sensitive. C. UV monitoring can also provide melting curve analysis, as illustrated in the panel.
UV real-time monitoring of amplicon production. Given that we use an all-optical approach to PCR, we continued to invest in assessing amplicon production in real- time optically. We have now successfully incorporated real-time monitoring of amplicon production using a UV label-free approach. The sensitivity of our plasmonic PCR is 10 DNA copy molecules, keeping on par with other diagnostic platforms. Our Plasmonic PCR or RT-PCR is now inclusive, completing and interpreting PCR in real time without added labels of any sort (i.e., label-free).
3. Hands and Chemical Free Sample Prep

Sample Preparation and Sterilization. Gold nanoparticles can generate significant bulk-heating of the reaction mixture to subsequently lyse bacteria and Clostridium spores and indeed we have demonstrated so. This can be extremely important regarding exposure risk to health workers.
4. Multiple Channels
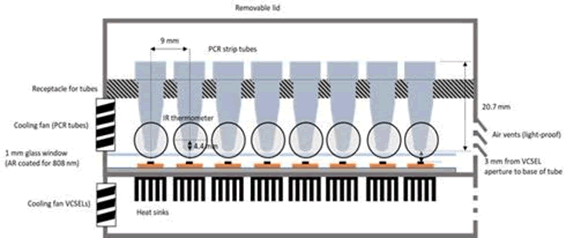
FIG 3. Up to 8-channels (4-channel instrument is shown) may be processed concurrently
5. Integration with Existing PCR, RT-PCR, qPCR
PCR Biology. With respect to polymerase enzymes, many enzymes and have found they all work within our platform. Our patent-approved Plasmonic PCR platform has been used to run all types of nucleic acid amplification protocols including PCR (Chlamydia/Gonorrhea), RT-PCR (HIV), and loop-mediate amplification (LAMP - Chlamydia/Gonorrhea). Our current effort is for the detection of COVID-19, hands free, label free in under one minute.
6. Point of Care

Systems is 18cm x 18cm x 18cm, 3kg with battery for an operating time of four hours. A true Point of Care system.
7. Reagents
Gold Nanorods (GNRs) from Nanopartz. Typically, the GNRs simply need to be added to the PCR mixture, with no other preparations required. More recently, we have developed room temperature stable lyophilized GNRs that are readily resuspended in water. This is very critical as one can contemplate the plasmonic PCR device without any concerns related to refrigeration requirements; as all components of PCR mixtures are also available lyophilized the POC implementation is now easier.
_______________________________________________________________________________________________________________________________
See our publications:
Real time label-free monitoring of plasmonic polymerase chain reaction products Gideon Uchehara, Andrew G. Kirk, Mark Trifiro, Miltiadis Paliouras, Padideh Mohammadyousef, Proc. SPIE 10969, Nano-, Bio-, Info-Tech Sensors and 3D Systems III, 109690A (27 March 2019); doi: 10.1117/12.2518295
Real time plasmonic qPCR: how fast is ultra-fast? 30 cycles in 54 seconds Roche, Philip J. R. ; Najih, Mohamed ; Lee, Seung S. ; Beitel, Lenore K. ; Carnevale, Matthew L. ; Paliouras, Miltiadis ; Kirk, Andrew G. ; Trifiro, Mark A. Analyst, 2017, Vol.142(10), pp.1746-1755
_________________________________________________________________________________________________________________________________
What does PCR in under 2 minutes look like:
_________________________________________________________________________________________________________________________________
For reagents contact us or order below!
