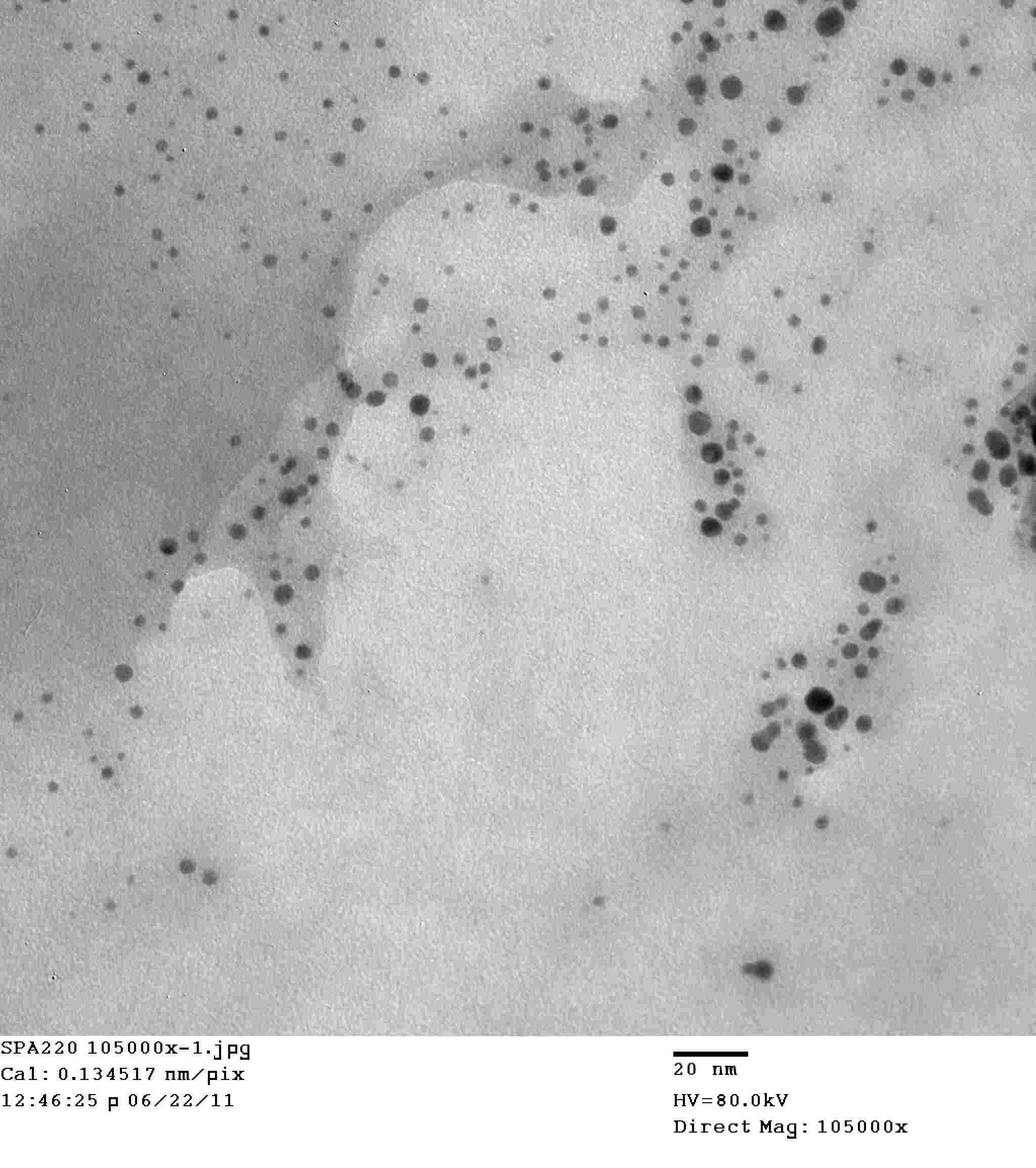
All About Cesium Oxide Nanoparticles
Definition, Properties and Applications
Cesium oxide nanoparticles (Cs2O NPs) are a type of nanomaterial with unique properties and various applications. Cesium oxide nanoparticles have been studied extensively due to their potential in different fields such as environmental remediation, catalysis, and biomedical applications. The properties and applications of cesium oxide nanoparticles are of significant interest due to their potential impact on various industries and environmental processes.
Cesium oxide nanoparticles have been investigated for their potential applications in environmental remediation. Studies have shown that nanocomposites containing Prussian blue type nanoparticles covalently linked to a matrix are promising for the decontamination of cesium ions (Delchet et al., 2012). Additionally, cesium-promoted ruthenium nanoparticles supported on multi-walled carbon nanotubes have been shown to be highly active for hydrogen production by ammonia decomposition, indicating potential applications in sustainable energy production (Hill & Torrente-Murciano, 2014). Furthermore, cesium oxide has been found to play a role in the side-chain alkylation of toluene with methanol, demonstrating its potential in catalytic processes (Han et al., 2016).
In the field of biomedical applications, cesium oxide nanoparticles have shown promise in various areas. For instance, cesium oxide nanoparticles have been used for the removal of cesium from human blood, demonstrating improved biocompatibility and potential for medical applications (Qian et al., 2017). Additionally, cesium nanoparticles have been investigated for their anti-cancer properties, showing potential for nanoparticle-mediated internalization and treatment (Daza et al., 2016).
The unique properties of cesium oxide nanoparticles, such as their low work function, make them of interest in the photodetection industry (Band et al., 2004). Furthermore, the stability of Cs2O closed cages has been highlighted, indicating potential applications in photoemissive devices and catalytic converters (Albu‐Yaron et al., 2005).
In summary, cesium oxide nanoparticles exhibit diverse properties and have a wide range of potential applications, including environmental remediation, catalysis, biomedical applications, and the photodetection industry. The unique properties of cesium oxide nanoparticles make them a subject of extensive research and hold promise for various practical applications.
Go here for Nanopartz Cesium Oxide Nanoparticles
References:
Albu‐Yaron, A., Arad, T., Popovitz‐Biro, R., Bar‐Sadan, M., Prior, Y., Jansen, M., … & Tenne, R. (2005). Preparation and structural characterization of stable cs2o closed‐cage structures. Angewandte Chemie, 44(27), 4169-4172. https://doi.org/10.1002/anie.200500356
Band, A., Albu‐Yaron, A., Livneh, T., Cohen, H., Feldman, Y., Shimon, L., … & Tenne, R. (2004). Characterization of oxides of cesium. The Journal of Physical Chemistry B, 108(33), 12360-12367. https://doi.org/10.1021/jp036432o
Daza, E., Misra, S., Schwartz-Duval, A., Ohoka, A., Miller, C., & Pan, D. (2016). Nano-cesium for anti-cancer properties: an investigation into cesium induced metabolic interference. Acs Applied Materials & Interfaces, 8(40), 26600-26612. https://doi.org/10.1021/acsami.6b09887
Delchet, C., Tokarev, A., Dumail, X., Toquer, G., Barré, Y., Guari, Y., … & Grandjean, A. (2012). Extraction of radioactive cesium using innovative functionalized porous materials. RSC Advances, 2(13), 5707. https://doi.org/10.1039/c2ra00012a
Han, H., Liu, M., Ding, F., Wang, Y., Guo, X., & Song, C. (2016). Effects of cesium ions and cesium oxide in side-chain alkylation of toluene with methanol over cesium-modified zeolite x. Industrial & Engineering Chemistry Research, 55(7), 1849-1858. https://doi.org/10.1021/acs.iecr.5b04174
Hill, A. and Torrente-Murciano, L. (2014). In-situ h2 production via low temperature decomposition of ammonia: insights into the role of cesium as a promoter. International Journal of Hydrogen Energy, 39(15), 7646-7654. https://doi.org/10.1016/j.ijhydene.2014.03.043
Qian, J., Xu, J., Kuang, L., & Hua, D. (2017). Cesium removal from human blood by poly(ethylene glycol)‐decorated prussian blue magnetic nanoparticles. Chempluschem, 82(6), 888-895. https://doi.org/10.1002/cplu.201700183