Customer Spotlight - Plasmonic Gold Nanostar Enhanced Multimodal Photoacoustic Microscopy and Optical Coherence Tomography Molecular Imaging
For the first time, scientists from the University of Michigan have developed an imaging technique that can visualize newly developed blood vessels in the back of the eye, named choroidal neovascularization (CNV).
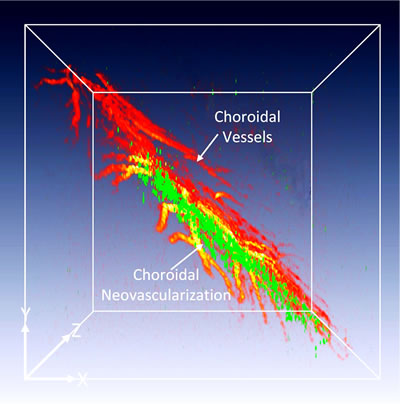
Merge three-dimensional photoacoustic microscopy of choroidal neovascularization (CNV) obtained from two different excitation wavelength of 578 nm (pseudo-green color) and 650 nm (pseudo-green color) |
Overview
While photoacoustic microscopy (PAM) and optical coherence tomography (OCT) allow visualization of the retinal microvasculature, distinguishing early neovascularization from adjacent vessels remains challenging. Herein, gold nanostars (GNS) functionalized with RGD peptide were utilized as multimodality contrast agents for both PAM and OCT. GNS have great absorption and scattering characteristics in the near-infrared region where most vasculature and tissue generates less intrinsic photoacoustic signal while having a small size, excellent biocompatibility in vivo, and great photostability under nanosecond pulsed laser illumination. This enabled visualization and differentiation of individual microvasculature in vivo using multimodal PAM and OCT imaging. Detailed three-dimensional imaging of GNS was achieved in an important choroidal neovascularization (CNV) model in living rabbits. Through the administration of GNS, PA contrast increased up to 17-fold and OCT intensity increased 167%. This advanced molecular imaging platform with GNS provides a unique tool for detailed mapping of the pathogenesis of the microvasculature.
Problem Addressed
Currently, there is no available imaging techniques can visualize newly developed blood vessels in the back of the eye, named choroidal neovascularization (CNV). Our group has developed a multimodal photoacoustic microscopy (PAM) and optical coherence tomography (OCT) to visualize normal and abnormal retinal vessels. However, hemoglobin has very low optical absorption and scattering in near-infrared window (650 to 1100 nm). Therefore, exogenous contrast agents can be used to enhance signal to noise ratio to allow one to distinguish CNV from adjacent vessels. This can allow for early diagnosis of retinal diseases, improved treatment, and reduced vision impairment.
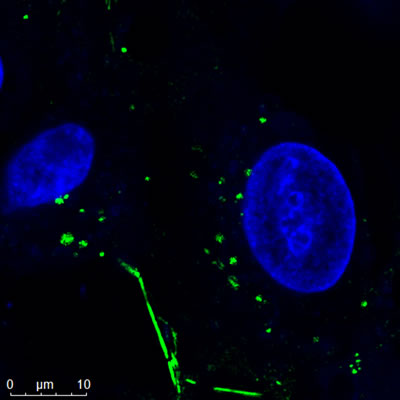
Confocal Image of Nanopartz Gold Nanorods |
Solution
The aim of our study is to apply gold nanoparticles (GNPs) conjugation with RGD peptides for enhanced PAM and OCT image contrast, resulting in improved visualization of CNV. The targeting ligands will allow GNPs bind to integrin αvβ3 which is overexpressed in CNV. Thus, the margin of CNV can be easily identify using spectroscopic PAM imaging.
Novelty, Applications, and Future work
The novelty of the system is the first multimodal PAM and OCT equipment that can visualize abnormal blood vessels in the back of the eye in large animal (rabbits) which has the eyeball size that is quite similar to that of the human eyeball axial size, leading to the ability to transfer this technique to clinical human applications. Prior to clinical use, there are some additional steps that are necessary. PAM imaging and GNPs are not approved by the US FDA for clinical use in these applications. A comprehensive safety evaluation should be performed to evaluate any potential harm to the eye. In the future, we would like to test smaller sized GNPs to reduce long term toxicity concern after administration.
Applications of this technology include GNPs to evaluate retinal diseases such as retinal neovascularization, CNV, and labeling of stem cells for regenerative therapy.
Currently, we are working on several projects to detect and treat retinal diseases and develop stem cells therapy. We are happy to collaborate with Nanopartz to apply different kinds of nanoparticles that can be used as theranostic agents.
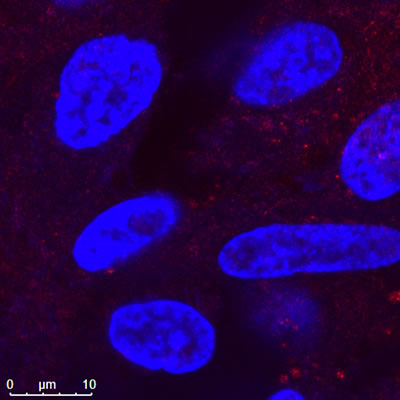
Confocal Image of Nanopartz Hollow Gold Nanoshells |
Nanopartz' Role
The nanoparticles from Nanopartz are biocompatible and stable with no toxicity at the tested concentrations. These GNPs can help to improve PAM and OCT image contrast after intravenous administration into the animal.
University of Michigan Contact and Publication
Yannis Mantas Paulus, MD, FACS Assistant Professor, Department of Ophthalmology and Visual Sciences Assistant Professor, Department of Biomedical Engineering Dr. Paulus' research seeks to allow physicians to diagnose diseases earlier, improve treatment monitoring, and practice more individualized precision medicine tailored to each patient through molecular imaging. My interest is in applying physics, bioengineering, and mathematical modeling to develop novel retinal imaging systems and laser therapies. Dr. Paulus Professional Page
Nguyen, Van Phuc, Yanxiu Li, Jessica Henry, Wei Zhang, Michael Aaberg, Sydney Jones, Thomas Qian, Xueding Wang, and Yannis Mantas Paulus. "Plasmonic gold nanostar-enhanced multimodal photoacoustic microscopy and optical coherence tomography molecular imaging to evaluate choroidal neovascularization." ACS sensors (2020). https://dx.doi.org/10.1021/acssensors.0c00908
Nanopartz Products Used for this Research
The products used for this research are our Gold Nanoparticles, Nanopartz Gold Nanorods, and Hollow Gold Nanoshells.